In this webinar presented by Dr. Neal Fischbach, we discuss the importance of PTEN alterations and how copy number loss can impact patient care.
Peer to Peer Education
Advanced Breast Cancer

Why Comprehensive Genomic Profiling (CGP) is Important in HR+/HER2- Advanced Breast Cancer
In this brief webinar presented by Dr. Lee Schwartzberg, we review current guidelines pertaining to CGP and biomarker-driven therapies for patients with advanced breast cancer.

Leveraging Genomics to Inform Care for Patients with Advanced Breast Cancer
Comprehensive genomic profiling (CGP) has become a powerful tool in oncology, enabling clinicians to identify genomic alterations that can guide targeted therapy options for patients. This approach is especially crucial for the 70% of advanced breast cancers classified as hormone receptor-positive (HR+) and human epidermal growth factor receptor 2 negative (HER2-). As the therapeutic landscape for these patients expands, the role of CGP continues to grow in significance.

Utilizing Tissue vs. Liquid Comprehensive Genomic Profiling (CGP) in Advanced Breast Cancer
In this brief webinar presented by Dr. Neal Fischbach, we review differences between tissue and liquid biopsy based CGP testing, including the appropriate time during the patient journey for each, and how to use ctDNA tumor fraction in clinical decision making for the advanced breast cancer patient with a driver negative liquid biopsy.

Germline vs. Somatic Alterations in Advanced Cancers
In this brief webinar presented by Dr. Lee Schwartzberg, we discuss the differences between genetic and genomic testing and alterations that are germline or somatic in origin.
Advanced Non-Small Cell Lung Cancer (NSCLC)

Why Comprehensive Genomic Profiling (CGP) is Important in Advanced NSCLC
In this brief webinar presented by Dr. Lee Schwartzberg, we review current guidelines pertaining to CGP and biomarker-driven therapies for patients with advanced non-small cell lung cancer (NSCLC).

Utilizing DNA and RNA in Advanced NSCLC
In this brief webinar presented by Dr. Todd Bauer, we review the benefits and limitations of DNA and RNA sequencing technologies and the impact on the care of patients with advanced non-small cell lung cancer (NSCLC).
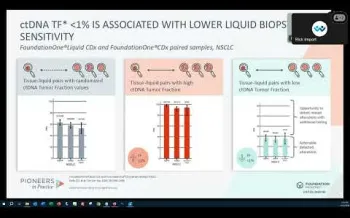
Understanding ctDNA Tumor Fraction in Advanced NSCLC
In this webinar presented by Dr. Hatim Husain, we review how to use ctDNA tumor fraction in clinical decision making for the advanced non-small cell lung cancer (NSCLC) patient with a driver negative liquid biopsy.

A Brief Review of DNA and RNA in Advanced NSCLC with Dr. Todd M. Bauer
In this brief 5-minute video, Dr. Todd M. Bauer reviews the history of next-generation sequencing for targeted therapies in advanced non-small cell lung cancer and discusses the differences between DNA and RNA-based sequencing technologies for these patients.
Advanced Prostate Cancer

Why is CGP important? A Pioneers in Practice Webinar
In this educational webinar, Dr. Scott Sellinger discusses why comprehensive genomic profiling (CGP) is important in advanced prostate cancer. He reviews pertinent guidelines and FDA-approved biomarker driven therapies for patients with advanced prostate cancer and briefly discusses the differences between genetic and genomic testing for these patients.

Tissue vs. Liquid CGP: A Pioneers in Practice Webinar
In this educational webinar, Dr. Jahan Aghalar discusses the differences between tissue and liquid biopsy comprehensive genomic profiling (CGP) in advanced prostate cancer. He reviews the benefits and limitations of tissue and liquid biopsy based CGP, discusses what circulating tumor DNA (ctDNA) is in comparison to all cell free DNA (cfDNA), and lastly introduces the concept of ctDNA tumor fraction pertinent to liquid biopsy based CGP.

Understanding ctDNA Tumor Fraction: A Pioneers in Practice Webinar
In this educational webinar, Dr. Jahan Aghalar reviews what ctDNA tumor fraction is and how it can help inform clinical decision-making in advanced prostate cancer. He reviews the benefits and limitations of tissue and liquid biopsy based CGP, defines what ctDNA tumor fraction is, how ctDNA tumor fraction is reported and lastly how it can impact clinical decision-making with uninformative negative liquid biopsy results.
ctDNA Tumor Fraction

ctDNA Revolution Shapes the Future of Cancer Care
Foundation Medicine has been at the forefront of liquid biopsy (LBx) advancements for nearly a decade, beginning with the launch of FoundationACTTM in 20161, a laboratory-developed test that marked Foundation Medicine’s entry into blood-based genomic profiling.

Review of Foundation Medicine's Biomarker, ctDNA Tumor Fraction & Implications in Clinical Practice
In this brief 4-minute video, Dr. Hatim Husain reviews the importance of utilizing a liquid biopsy test that reports ctDNA tumor fraction with a validated cut-off to guide clinical decision making, and how it has made an impact in his practice and with his patients.
Meet Our Experts

Jahan Aghalar, MD
Dr. Jahan Aghalar is a community-based oncologist with over a decade of experience who focuses on the treatment of genitourinary cancers, such as advanced prostate cancer. Dr. Aghalar is an active member of both the American Society of Hematology (ASH) and the American Society of Clinical Oncology (ASCO), and also serves on multiple committees such as the OneOncology GU Disease Group and Educational Committees, the Castrate Resistant Prostate Cancer Quality Improvement Committee and he chairs the NCCN’s Health Equity Report Card for Community Settings, among others. Dr. Aghalar is passionate about educating others on the importance of comprehensive genomic profiling in helping to find answers that guide treatment decisions for patients with advanced prostate cancer.

Todd Bauer, MD
Dr. Todd Bauer is an oncologist currently with over 15 years of experiences in treating patients with cancer. He currently practices in the community setting in Nashville, Tennessee. Dr. Bauer is an active member of multiple medical societies and holds numerous committee and administrative assignments. He impressively has over 85 publications, many of which involve biomarker discovery and targeted therapeutics, and has presented numerous abstracts and oral presentations and at large societal conferences. Dr. Bauer is an expert in the space of thoracic oncology and biomarker-driven therapy and is passionate about providing precision oncology care for his patients.

Neal Fischbach, MD
Dr. Neal Fischbach is an oncologist currently with nearly three decades of experience in treating patients with cancer. He currently practices in the academic medical setting. Dr. Fischbach is passionate about ensuring all patients have equitable access to clinical trials, and also generously gives his time and expertise in working with non-profits serving patients with cancer. He is a member of multiple medical societies and committees and has over 20 peer reviewed publications. Dr. Fischbach is an expert at treating patients with advanced breast cancer and providing personalized cancer.

Hatim Husain, MD
Dr. Hatim Husain is a thoracic oncologist and biomarker expert. He has over a decade of experience practicing in the academic medical setting and is a member of multiple medical societies and committee. While Dr. Husain has a multitude of peer-reviewed publications, he specifically has been a senior author on multiple ctDNA tumor fraction related papers pertinent to liquid biopsy. Dr. Husain has also given numerous scientific presentations and participated clinical trials over his career. He is passionate not only about precision oncology care but also about utilizing ctDNA tumor fraction for clinical decision-making in advanced lung cancer.

Lee Schwartzberg, MD
Dr. Lee Schwartzberg is an oncologist with over 4 decades of experience in treating patients with cancer. He currently practices in community oncology and also holds an academic teaching appointment. Dr. Schwartzberg is an active member of multiple medical societies and committees and has received numerous honors and awards for his extensive work with the oncology community. He also has over 300 peer reviewed publications and has given numerous presentations. Dr. Schwartzberg is an expert in treating patients with advanced breast cancer and advanced lung cancer and is passionate about providing optimized personalized medicine.

Scott Sellinger, MD
Dr. Scott Sellinger is a urologist with over 4 decades of experience in community urology practice. He is the President of a large independent community practice in Tallahassee, Florida as well as a member of multiple medical societies and committees. His focus is on treating patients with advanced prostate cancer. Dr. Sellinger has a passion for educating others on this topic and for ensuring patients have access to biomarker driven therapies.
Additional Notes
FoundationOne®CDx and FoundationOne®Liquid CDx are qualitative next-generation sequencing based in vitro diagnostic tests for advanced cancer patients with solid tumors and are for prescription use only. FoundationOne CDx utilizes FFPE tissue and analyzes 324 genes as well as genomic signatures. FoundationOne Liquid CDx analyzes 324 genes utilizing circulating cell-free DNA and is FDA-approved to report short variants in 311 genes. The tests are companion diagnostics to identify patients who may benefit from treatment with specific therapies in accordance with the therapeutic product labeling. Additional genomic findings may be reported and are not prescriptive or conclusive for labeled use of any specific therapeutic product. Use of the tests does not guarantee a patient will be matched to a treatment. A negative result does not rule out the presence of an alteration. Some patients may require a biopsy for testing with FoundationOne CDx when archival tissue is not available which may pose a risk. When considering eligibility for ROZLYTREK® based on the detection of NTRK1/2/3 and ROS1 fusions, or for TEPMETKO® based on the detection of MET SNVs and indels that lead to MET exon 14 skipping, testing using plasma specimens is only appropriate for patients for whom tumor tissue is not available for testing. Patients who are tested with FoundationOne Liquid CDx and are negative for other companion diagnostic mutations should be reflexed to tumor tissue testing and mutation status confirmed using an FDA-approved tumor tissue test, if feasible. For the complete label, including companion diagnostic indications and important risk information, please visit www.F1CDxLabel.com and www.F1LCDxLabel.com.